Description
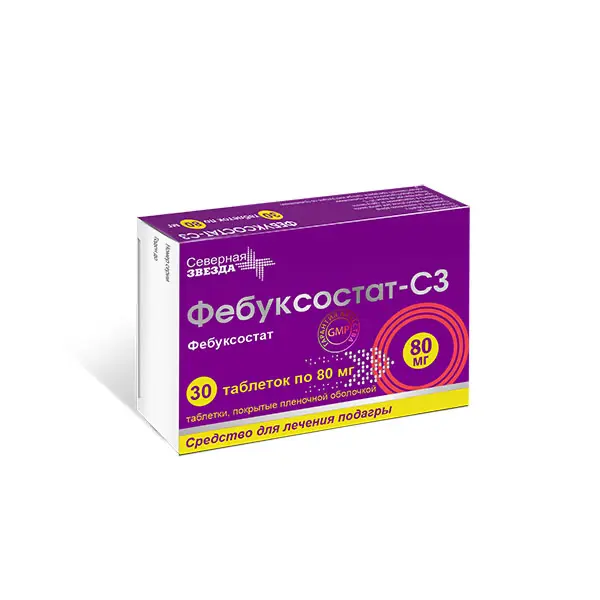
Febuxostat-SZ Pharmacodynamics
Uric acid is a final product of purine metabolism in human body, formed as a result of hypoxanthine-xanthine uric acid reaction cascade. Febuxostat is a 2-arylthiazole derivative and is a strong selective non-purine xanthine oxidase inhibitor (in vitro inhibition constant is less than 1 nM). The enzyme xanthine oxidase catalyzes two stages of purine metabolism: oxidation of hypoxanthine to xanthine and then oxidation of xanthine to uric acid.
As a result of selective inhibition of xanthine oxidase (oxidized and reduced forms) by febuxostat there is a decrease in the concentration of uric acid in blood plasma.
At therapeutic concentrations febuxostat does not inhibit other enzymes involved in the metabolism of purines or pyrimidines, such as guanine deaminase, hypoxanthineguanine phosphoribosyltransferase, orotate phosphoribosyltransferase, orotidine monophosphate decarboxylase or purine nucleoside phosphorylase.
Indications
Treatment of chronic hyperuricaemia in conditions accompanied by deposits of urate crystals (in the presence of tophi and/or gouty arthritis, including in the anamnesis).
The drug Febuxostat-SZ is intended for use in adults.
Contraindications
– Hypersensitivity to Febuxostat and/or any of the auxiliary substances;
– severe hepatic insufficiency class C according to Child-Pugh scale (10-15 points);
– severe renal insufficiency (creatinine clearance <30 ml/min);
– children under 18 years of age;
– Pregnancy and breastfeeding;
– hereditary galactose intolerance, lactase deficiency and glucose and galactose malabsorption syndrome.
Dosage and administration
- Orally. Preparation Febuxostat-SZ is taken once a day, regardless of meals.
- Gout
- Recommended starting dose is 80 mg of Febuxostat once daily regardless of meals. In case plasma uric acid concentration exceeds 6 mg/dL (357 µmol/L) after 2-4 weeks the dose can be increased up to 120 mg once a day.
- Decrease of uric acid concentration in blood plasma against the background of using Febuxostat-SZ occurs rather quickly, and therefore control of uric acid concentration can be performed after two weeks from the start of the drug. The aim of the treatment is to decrease and maintain uric acid concentration in blood plasma less than 6 mg/dl (357 µmol/l).
- Prevention of the development of acute attacks of gout is recommended for at least 6 months.
- Tumor decay syndrome.
- The recommended dose is 120 mg of Febuxostat once daily regardless of meals. Febuxostat-SZ should be started two days before the start of cytostatic chemotherapy. The duration of Febuxostat-SZ administration should be at least 7 days. However, the duration of therapy can be increased up to 9 days, depending on the duration of chemotherapy and clinical evaluation.
- Elderly patients.
- No dose adjustment of the drug is required.
- Patients with hepatic insufficiency
- Study of febuxostat efficacy and safety in patients with hepatic insufficiency of severe degree (grade C according to Child-Pugh scale (10-15 points)) have not been carried out, due to this fact the preparation use in such patients is contraindicated.
- Gout
- Recommended dose for patients with mild hepatic insufficiency (grade A according to Child-Pugh scale (5-6 points)) is 80 mg 1 time per day. The experience of using the drug in mild hepatic insufficiency is limited.
- Tumor decay syndrome.
- In patients participating in FLORENCE trial no dose adjustment of febuxostat depending on liver function was required (patients with severe hepatic insufficiency were not included in the study).
- Patients with renal failure
- In patients with mild to moderate renal failure no dose adjustment is required.
- In patients with severe renal failure (creatinine clearance <30 ml/min) efficacy and safety of the drug have been insufficiently studied, therefore, the drug is contraindicated in such patients.