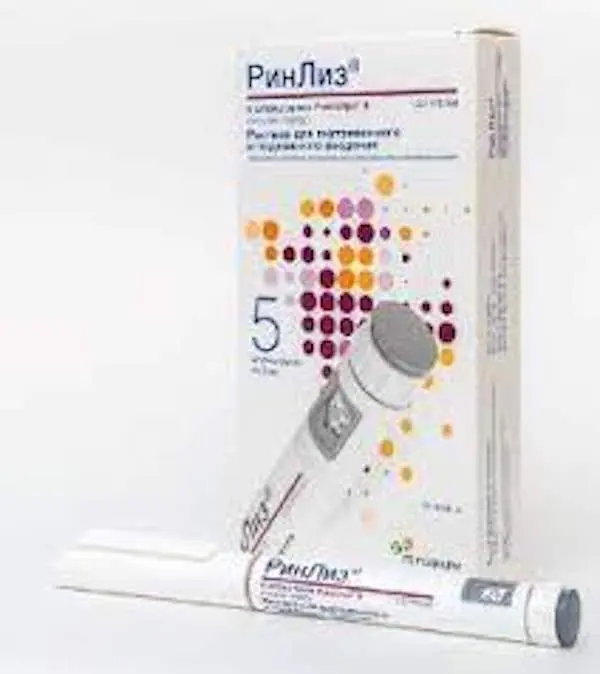
Description
RinLys Pharmacodynamics
RinLys® is a DNA recombinant analogue of human insulin. It differs from human insulin by the reverse sequence of amino acids at positions 28 and 29 of the B-chain of insulin.
Pharmacodynamics
The main action of insulin lispro is regulation of glucose metabolism.
In addition, it has anabolic and anti-catabolic effect on different tissues of the body. In muscle tissue there is an increase in glycogen, fatty acids, glycerol content, increased protein synthesis and increased consumption of amino acids, but there is a decrease in glycogenolysis, gluconeogenesis, ketogenesis, lipolysis, protein catabolism and release of amino acids.
Insulin lispro has been shown to be equimolar to human insulin, but its action is more rapid and shorter in duration. Insulin lispro is characterized by a rapid onset of action (around 15 minutes), as it has a high speed of absorption, and this allows it to be injected immediately before a meal (0-15 minutes before the meal), in contrast to the usual short-acting insulin (30-45 minutes before the meal). Insulin lispro is fast-acting and has a shorter duration of action (2 to 5 hours) compared to human insulin.
Patients with type 1 and type 2 diabetes with insulin lispro have a more pronounced reduction in postprandial hyperglycemia compared to soluble human insulin.
As with all insulin preparations, the duration of action of insulin lispro may vary in different patients or at different times in the same patient and is dependent on dose, site of injection, blood supply, body temperature, and physical activity.
The pharmacodynamic characteristics of insulin lispro in children and adolescents are similar to those seen in adults.
Insulin lispro in patients with type 1 and type 2 diabetes mellitus has a lower incidence of nocturnal hypoglycemia episodes compared to soluble human insulin.
The glucodynamic response to insulin lispro is not dependent on renal or hepatic function.
Indications
Diabetes mellitus in adults and children requiring insulin therapy to maintain normal blood glucose levels.
Contraindications
– Hypersensitivity to insulin lispro or to any of the drug components;
– Hypoglycemia.
Usage during pregnancy and breast-feeding.
Pregnancy
Numerous data on the use of insulin lispro during pregnancy indicate no adverse effects of insulin lispro on pregnancy and on the fetus and newborn.
During pregnancy, the primary goal of insulin therapy is to maintain good glycemic control in insulin-treated diabetic patients. The need for insulin usually decreases during the first trimester and increases during the second and third trimesters of pregnancy. During and immediately after childbirth, the need for insulin may decrease dramatically. Patients with diabetes should consult a physician if they become pregnant or are planning to become pregnant. In the case of pregnancy in patients with diabetes, careful monitoring of glucose as well as general health is essential.
Breastfeeding
Patients who are breastfeeding may need to adjust their insulin dosing regimen, diet, or both.
Dosage and administration method
- Dosage of RinLiz® is determined by a physician individually, depending on blood glucose concentration. Mode of insulin administration is individual.
- RinLys® may be administered immediately before meals. If necessary, RinLiz® may be administered immediately after the meal.
- The temperature of the administered drug must correspond to room temperature.
- For RinLysis® in cartridges
- RhinLys® should be administered by subcutaneous injections or by prolonged subcutaneous infusion using an insulin pump. If necessary (ketoacidosis, acute diseases, inter-operative period or postoperative period), RinLiz® can also be administered intravenously.
- For RinLysis® in a Rinastra® II or Rinastra® III disposable syringe pen
- Rinastra® II or Rinastra® III syringe pens are only suitable for subcutaneous injections.
- Subcutaneous injections should be given in the upper arm, thigh, buttock, or abdomen. The injection sites should be alternated so that the same site is not used more than about once a month.
- When injecting RinLys® subcutaneously, care must be taken to avoid getting the drug into a blood vessel. The injection site should not be massaged after the injection. The patient should be trained in the correct injection technique.
- RinLys® may be used under control of a physician in combination with longer-acting insulin or in combination with oral hypoglycemic preparations.
- Use in children and adolescents.
- RinLiz® may be used in children and adolescents.
- Instructions for administration of RinLiz®
- Preparation for drug administration
- RhinLys® solution must be clear and colourless. Do not use if the solution is cloudy, thickened or poorly coloured, or if solid particles are visually detected.
- When inserting the cartridge into the syringe pen, attaching the needle, and injecting the insulin, follow the manufacturer’s instructions that come with each syringe pen.
- Dose administration
1. Wash your hands.
2. Select a location for the injection.
3. Prepare the skin at the injection site according to the doctor’s recommendations.
4. Remove the outer protective cap from the needle.
5. Secure the skin by pulling it taut or gathering it in a large fold.
6. Insert the needle subcutaneously and inject according to the syringe pen instructions.
7. Remove the needle and gently press the injection site with a cotton swab for a few seconds. Do not rub the injection site.
8. Using the outer needle protector cap, unscrew the needle and dispose of it.
9. Put the cap back on the syringe pen.
- Intravenous administration of insulin
- Intravenous injections of RinLiz® should be given in accordance with routine clinical practices for intravenous injections, such as intravenous bolus or intravenous infusion using an infusion system. This requires frequent monitoring of blood glucose concentrations.
- Infusion systems with concentrations of 0.1 IU/ml and up to 1.0 IU/ml of insulin lispro in 0.9% sodium chloride solution or 5% dextrose solution are stable at room temperature for 48 hours.
- Subcutaneous insulin infusion using an insulin pump
- CE-marked continuous subcutaneous insulin pump systems may be used for infusion of RinLys®. Before administering the insulin lispro, you must check the suitability of the pump in question. The instructions provided with the pump must be strictly followed. Use a suitable reservoir and catheter for the pump. The infusion set should be used according to the instructions supplied with the infusion set. If a hypoglycemic reaction develops, the infusion is stopped until the episode resolves. If very low blood glucose concentrations are noted, this should be reported to the physician and provision made to reduce or stop the insulin infusion. Malfunction of the pump or clogging of the infusion system can lead to a rapid rise in blood glucose levels. If insulin supply failure is suspected, follow the instructions and inform your physician if necessary. When using the pump, RinLys® should not be mixed with other insulins.